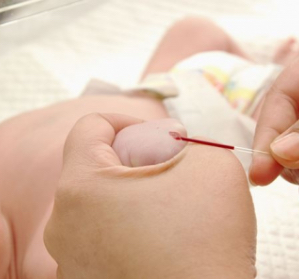
Early detection programmes for rare diseases, which in some countries are already 50 years old, aim to reduce the morbidity and mortality and disabilities associated with these pathologies. There is no worldwide unanimity on the diseases to be detected; mainly metabolic processes are identified, but also endocrine, haematological, immunological and cardiological processes. Neonatal screening for rare diseases received a major boost in the 2000s, with the use of tandem mass spectrometry to determine acylcarnitines and amino acids in a paper-impregnated blood sample. The introduction of emerging technologies applicable to new biochemical biomarkers and DNA analysis by mass sequencing may have a favourable impact on the future of rare disease screening. Therefore, an agile decision-making system is needed to assess the inclusion of new pathologies in neonatal screening.